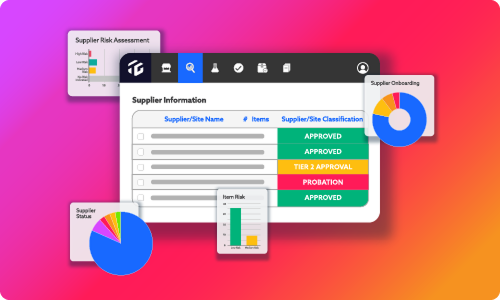
DIGEST
The 7-Step Guide to Packaging Specification Management for Food & Beverage Innovation
How a connected spec ecosystem aligns quality, R&D, packaging, and the business.

THE PROBLEM
Why specs are now a business system
For decades, specifications were treated as supporting documents. Necessary, but secondary to the “real work” of product development, sourcing, and launch execution. Formulas lived with R&D. Packaging specs sat with packaging teams. Artwork moved through marketing workflows. Compliance documentation was assembled when needed.

That approach no longer works.
Today, food and beverage innovation operates in a fundamentally different environment. Product portfolios are more complex. Supplier networks are more distributed. Regulatory expectations are higher. Artwork and labeling requirements vary by market. And innovation cycles continue to shrink.
In this reality, specifications are no longer just records of what was decided. They are the system that determines whether teams can move forward with confidence.
When specs are disconnected across teams and tools, the impact is felt everywhere.
Hover over the ghosts to uncover the hidden pitfalls of a disconnected supply chain.

Lost hours
R&D slows down as teams revalidate information

Misaligned data
Packaging and artwork fall out of sync

Missing trails
Quality and Regulatory scramble to assemble audit-ready documentation

Blind decisions
Procurement struggles to align sourcing with approved specs

Missed launches
Marketing and Sales face delays and last-minute changes

Broken systems
IT supports a patchwork of tools that were never designed to work together

Avoidable costs
Finance absorbs the cost of rework, delays, and avoidable risk
What looks like a documentation problem is, in reality, a business system problem.
THE SOLUTION
Lost in the spec maze? There's an easy way out.
Packaging Specification Management provides that connective layer. Not as another tool, but as a connected ecosystem that treats specifications as shared infrastructure across the organization.
A connected spec ecosystem brings formula, ingredient, packaging, finished goods, and artwork data into a single, structured lifecycle. It replaces manual handoffs with defined relationships. It ensures that when something changes, the right teams see it, review it, and act on it; without chaos.
In short, it turns specs into a business system that supports innovation, compliance, collaboration, and growth.
This guide walks through a practical, seven-step framework for adopting Packaging Spec Management in a way that aligns every major function — from Quality and R&D to Finance and IT — around a shared source of truth.
THE GUIDE
The 7-step framework
Packaging Spec Management changes the role of specifications by fundamentally changing how they’re structured, connected, and used across the organization. The following seven steps outline a practical path forward. One that organizations can adopt incrementally, while still delivering measurable value at each stage.
Each step addresses a common breakdown point in product development and shows how a connected spec ecosystem creates alignment across teams.
STEP 1
Centralize the source of truth
Bring formulas, ingredients, packaging, and finished goods into one trusted system so teams stop reconciling versions and start executing.
Problem: Specs scattered across spreadsheets, drives, emails, and supplier portals.
In most food and beverage (F&B) organizations, specifications don’t live in one place. They live everywhere. Formulas sit in R&D spreadsheets. Ingredient specs arrive as PDFs from suppliers. Packaging requirements hide in email threads. Finished goods documentation lives in shared drives or legacy systems no one fully trusts. Each system may be “right” in isolation, but together they create uncertainty.
The real risk isn’t missing data; it’s conflicting data. Teams waste time debating which version is current, re-validating work that’s already been done, or redoing it entirely when errors surface late. Audits become scavenger hunts. Innovation slows not because teams lack ideas, but because they lack confidence in the information behind them.
What changes when specs are connected
Centralizing the source of truth means establishing one authoritative system for formulas, ingredients, packaging, and finished goods specifications built on structured, connected data rather than static documents. Instead of managing dozens of files that must be manually updated and re-shared, teams work from a single, living dataset where changes cascade automatically to wherever they’re needed.
This shift is foundational. It turns specifications from records of work into infrastructure for work. Data becomes searchable, comparable, and reusable across products and projects. Supplier updates flow into the same environment as internal specs. Packaging, formulation, and compliance data stay aligned by design, not by constant coordination.
How different teams benefit
- Quality & Regulatory: Fewer gaps between documents, clearer audit trails, and faster access to exactly what inspectors ask for, without manual reconciliation.
- R&D: Less rework caused by outdated inputs, faster iteration during formulation, and confidence that downstream teams are working from the same baseline.
- IT: Fewer one-off tools, fewer shadow systems, and a more governable architecture that supports scale without constant customization.
Outcome
When there is one trusted source of truth, teams stop asking “Is this the right version?” and start focusing on execution. Confidence in “what’s current” spreads across the organization, reducing friction, accelerating decisions, and setting the stage for true end-to-end connectivity.
STEP 2
Structure packaging specs across levels
Organize packaging across materials, components, and SKUs so changes are clear, consistent, and scalable.
Problem: Packaging data exists, but without hierarchy or clarity.
Even when packaging specifications are centralized, many organizations struggle with structure. Materials, components, assemblies, and SKU-level requirements are often documented independently, without clear relationships between them. The result is duplication, ambiguity, and manual interpretation, especially when changes occur.
A label update may not clearly roll up to the finished good. A material change might require downstream updates that teams must remember to make manually. Over time, inconsistencies creep in, and packaging becomes harder to manage as portfolios grow.
What changes when specs are structured
Structuring packaging specifications across levels introduces a clear, logical hierarchy: raw materials → packaging construction → SKU-level specifications. Each level has a defined role, ownership, and relationship to the others. Shared components are referenced, not copied. Changes propagate through the structure automatically, reducing the risk of missed updates or conflicting requirements.
This approach eliminates ambiguity by making packaging data modular and reusable. Teams no longer need to recreate specs for every SKU variation or manually reconcile differences across files. Instead, packaging becomes a connected system that reflects how products are actually built and produced.
How different teams benefit
- Packaging: Clear ownership of materials and assemblies, consistent specifications across SKUs, and fewer last-minute clarifications during execution.
- Procurement: Better alignment between sourcing decisions and approved packaging requirements, enabling smarter supplier engagement and fewer surprises.
- Finance: Reduced waste, fewer reprints, and less rework driven by misaligned or duplicated specs; costs that often hide until it’s too late.
Outcome
With structured, hierarchical packaging specifications, packaging stops being a manual coordination exercise. It becomes a manageable, scalable part of product development; one that supports speed, accuracy, and growth instead of constraining it.
STEP 3
Connect formulas, packaging & finished goods
Link all product data into finished goods so teams gain full visibility into what’s approved and production-ready.
Problem: Teams manage pieces, but finished goods lack full visibility.
Most organizations don’t struggle because they lack data. They struggle because the data lives in pieces. Formulas sit with R&D. Packaging specs live somewhere else. Artwork, supplier documentation, and market variations follow their own paths. Each system may work in isolation, but finished goods, the thing you actually manufacture and sell, rarely have a complete, connected view.
That gap shows up late, when it’s expensive.
When teams manage formulas, packaging, and SKUs separately, finished goods become an afterthought assembled mentally, not digitally. Questions like “Is this SKU production-ready?” or “Does this package reflect the approved formula?” require meetings, emails, and manual cross-checks. The result is uncertainty right where confidence matters most.
What changes with end-to-end clarity
Connected Packaging Spec Management treats assemblies and finished goods as the convergence point where everything comes together by design, not by guesswork.
Formulas, ingredients, packaging components, and labels roll up into structured assemblies that reflect how products are actually built. Those assemblies then support one-to-many relationships across SKUs, regions, and markets. A single packaging configuration can serve multiple finished goods, even as artwork or labeling changes by market.
Instead of duplicating specs or rebuilding context every time a product expands, teams reuse what’s already approved, intentionally and transparently.
How different teams benefit
- R&D: Gains confidence that formula updates won’t create downstream chaos. Changes flow into finished goods automatically, reducing rework and late-stage surprises.
- Operations: See production readiness clearly—what’s approved, what’s pending, and what’s actually buildable—without chasing information across systems.
- Sales & Marketing: Know that what’s being launched is real, approved, and aligned, making it easier to commit to timelines, promos, and customers.
Outcome
Finished goods stop being a blind spot. Teams gain end-to-end clarity, from raw materials to market-ready SKUs. Instead of asking whether everything lines up, they can see it instantly. This is where connected specs move from theory to execution.
STEP 4
Bring artwork into the spec lifecycle
Connect artwork directly to specs so labels, claims, and packaging stay aligned from approval to production.
Problem: Artwork approvals happen outside the spec process.
Even with well-managed specs, many product launches still fail at the same point: artwork. That’s because artwork approvals often live outside the product development system entirely, managed in separate tools, email threads or shared drives. Specs may change, but artwork doesn’t always keep up. And when misalignment surfaces, it’s usually late, visible, and costly.
Packaging artwork isn’t just creative; it’s regulatory, operational, and commercial. A single outdated ingredient statement, claim, or barcode can trigger reprints, delays, compliance risk, or worse. Yet without a direct connection to specs, teams rely on manual coordination to keep everything aligned.
What changes with artwork integration
When artwork is treated as part of the specification lifecycle, not a parallel process, alignment becomes automatic instead of manual.
Packaging specifications and finished goods are directly linked to artwork projects, creating a clear line of sight between what’s been approved and what’s being designed, reviewed, and released. Teams can instantly see which products have approved artwork, which are still in progress, and which changes require review without chasing emails or reconciling versions.
How different teams benefit
- Packaging & Marketing: Avoid last-minute fixes, rushed approvals, and emergency reprints by staying aligned earlier in the process.
- Quality & Compliance: Reduce labeling risk with clear traceability between approved specs and approved artwork.
- Supply chain: Gains better control over production timing, materials, and documentation across partners and markets.
Outcome
Artwork no longer lags behind product reality. It stays connected, current, and auditable so what gets printed matches what gets approved. And with that final piece in place, the spec lifecycle truly runs from concept to shelf.
STEP 5
Automate change control across teams
Turn change into a controlled process with automated workflows that track impact, trigger reviews, and prevent errors.
Problem: Change triggers chaos instead of coordination.
Change is inevitable in F&B product development. What isn’t inevitable is the confusion that follows it. A formula update. A supplier change. A packaging tweak. Each adjustment sets off a chain reaction across specs, artwork, approvals, and timelines. Without a connected system, teams rely on emails, spreadsheets, and memory to manage the fallout. Important reviews get missed. Outdated versions slip through. And issues surface late, when they’re expensive to fix.
In disconnected environments, change control is reactive. Teams discover updates after the fact, approvals happen out of sequence, and no one has a complete picture of what’s been impacted. The result isn’t just inefficiency, it’s risk. Risk of noncompliance. Risk of rework. Risk of delays that ripple across the business.
What changes with automated control
Automated change control replaces guesswork with structure.
When a spec changes, the system identifies what’s affected—ingredients, packaging components, assemblies, finished goods, and related artwork—and routes those changes through automated review and approval workflows. The right people are notified at the right time, with full context, instead of being pulled in through side conversations.
Version control becomes built in, not bolted on. Teams can see what changed, why it changed, and which version is current without reconciling files or second-guessing approvals. Nothing moves forward until required reviews are complete, and nothing gets lost in the shuffle.
How different teams benefit
- Quality: Gain confidence that no change slips through without proper review or documentation, reducing audit risk and compliance gaps.
- IT: Spend less time supporting manual workarounds, reconciling systems, or managing exceptions.
- Finance: Fewer costly errors, avoided reprints, reduced rush fees, and more predictable launch timelines.
Outcome
Change stops being a fire drill. It becomes a managed, transparent process that teams trust. With automated change control in place, organizations move faster not because they avoid change, but because they’re finally equipped to handle it.
STEP 6
Extend the ecosystem beyond internal teams
Collaborate with suppliers and partners in one system so everyone works from the same accurate, up-to-date data.
Problem: Supplier and partner collaboration breaks the chain of trust.
Even the most organized internal system breaks down when it hits the outside world.
Suppliers send documents in different formats. Co-manufacturers maintain their own versions of specs. Retailers request updates through email or portals that don’t sync with your records. Every handoff introduces friction, and every gap increases the chance that something outdated or incomplete slips through.
The result isn’t just inefficiency. It’s a break in trust.
What changes with an extended ecosystem
Connected Packaging Spec Management extends beyond internal users to create a shared, controlled ecosystem.
Specs and documents can be securely shared with suppliers, co-manufacturers, and retail partners directly within the platform. Instead of attachments floating between inboxes, everyone accesses the same structured, current information.
At the same time, ownership never gets lost. You maintain control over versions, permissions, and approvals, with clear audit trails showing who viewed, updated, or approved what and when.
This turns collaboration from a series of manual exchanges into a coordinated, transparent process.
Partners don’t just receive documents. They participate in a connected workflow.
How different teams benefit
- Procurement: Build stronger supplier relationships with faster onboarding, clearer expectations, and fewer back-and-forth corrections.
- FSQ team: Gain cleaner compliance documentation and instant access to certificates, specs, and supporting data.
- Operations: Fewer miscommunications that cause production delays or last-minute substitutions.
Outcome
Instead of disconnected partners operating on different versions of reality, you create a connected network built on shared data and shared trust.
Because your supply chain is only as strong as the information connecting it.
STEP 7
Turn specs into strategic advantage
Transform spec data into reusable intelligence that accelerates innovation, reduces risk, and scales growth.
Problem: Specs are treated as overhead, not assets.
For years, specifications have been treated like paperwork. Something you complete for compliance. Something you store for audits. Something you update when you have to.
But in a connected system, specs stop being static documents. They become structured, usable intelligence; the foundation that every team relies on to move faster and make better decisions.
When specs live in disconnected files, they slow everything down. Teams duplicate work. Leaders lack visibility. Every new product or market expansion feels heavier than it should. Innovation stalls, not because ideas are lacking, but because the operational lift is too high.
Specs feel like a cost center.
What changes with spec intelligence
When formulas, packaging, finished goods, and artwork are fully connected, your data becomes dynamic.
You can instantly see what’s approved, what’s reusable, what’s impacted by change, and what’s ready for launch. Teams reuse validated components instead of starting from scratch. Leaders spot risks earlier. And scaling into new markets or SKUs becomes a configuration exercise, not a documentation marathon.
This is spec intelligence: turning everyday operational data into insight that supports speed, scalability, and resilience.
Instead of asking, “Can we handle this?” teams ask, “How fast can we launch?”
How different teams benefit
- Executives & CFOs: Measurable ROI through fewer errors, faster billing, reduced waste, and lower risk exposure.
- Innovation leaders: Bring products to market faster without sacrificing accuracy or compliance.
- IT: Growth with a scalable, future-ready architecture rather than a patchwork of tools.
Outcome
Specs become more than records. They become a growth enabler.
Because when your foundation is connected, everything built on top of it moves faster.
And that’s when Packaging Spec Management shifts from operational improvement to strategic advantage.
CONCLUSION
One ecosystem, shared value
Where formulas, packaging, and artwork finally work as one. If there’s one takeaway from this journey, it’s simple:
- Product development doesn’t break down because of teams.
- It breaks down because the system connecting them doesn’t exist.
When formulas live in one place, packaging specs in another, finished goods somewhere else, and artwork in a separate workflow entirely, teams are forced to coordinate manually. Every launch becomes a series of handoffs. Every change becomes a risk. Specifications become paperwork instead of power.
The seven steps you’ve just walked through change that foundation.
-
- Centralize your source of truth.
- Structure packaging across clear levels.
- Connect formulas, packaging, and finished goods.
- Bring artwork into the lifecycle.
- Automate change control.
- Extend collaboration to partners.
- Turn spec data into intelligence.
Individually, these improvements save time. Together, they create something much bigger: a connected spec ecosystem that runs the business. That ecosystem is exactly what Packaging Specification Management delivers.
It isn’t a bolt-on feature or a single workflow. It’s a purpose-built TraceGains solution that brings together:
- Formula Management to digitize and control recipes
- Specification Management to structure items, ingredients, and materials
- Packaging Specification Management to manage components, assemblies, and SKU relationships
- Finished Goods to unify everything into production-ready products
- Integrated artwork approvals with Esko WebCenter Go to connect labels, claims, and creative directly to approved specs
So instead of juggling systems, teams operate inside one continuous lifecycle, from source to shelf.
R&D moves faster.
Packaging works with confidence.
Quality sleeps better at night.
All teams work from the same reality.
No more discovering misalignment at the last minute. Just shared visibility, coordinated workflows, and confidence that what’s approved is what gets produced. That’s what makes Packaging Specification Management different. It’s not another tool to manage. It’s infrastructure you build on.
And when that infrastructure is connected, every team moves faster.
Ready to meet Packaging Specification Management?
Explore Packaging Specification Management or request a demo to see how TraceGains and WebCenter Go bring your entire product lifecycle together.