More information isn’t always better; sometimes it’s just more. For teams already drowning in supply chain paperwork—digital or otherwise—a strong data collection strategy becomes far more valuable than additional data itself. But if you don’t know where to start when developing said strategy, asking several key questions can give you a firm foundation.
Access relevant, contextual data on demand
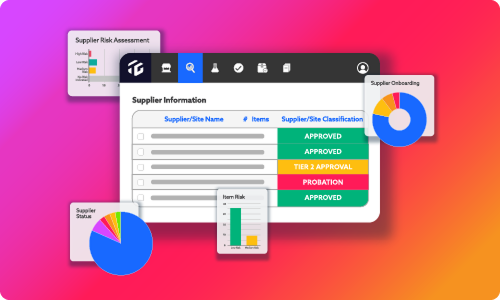
With the latest advancements in technology, there’s no shortage of solutions that can make information more accessible. Yet, how do you ensure that it’s relevant to your supply chain? If the goal is to save your team can save time searching for compliance details, supplier data, lot details, and more when it matters most, then these preliminary questions are essential to create contextual insights.
Be ready to interpret the results
Thinking beyond fancy reports and/or glossy graphs, your answers these seven questions can inform what “right” looks like within your organization. Not to mention how you evaluate success across all levels and teams. By deciding ahead of time which metrics are most meaningful, you can give your data-driven overhaul a head start and maximize the insights gained from your efforts.Ask the right questions
Have we piqued your interest? Simply complete the form and download this convenient infographic to see all seven questions now. If your team struggles with finding the right answers, contact us directly to learn more about how TraceGains can help.